LOUIS-PLENCE Pascale (CRHC, INSERM), ORCID
SHAH Nainisha (PhD UM)
FORTUIN Annemarie (PhD Inserm)
GARCIN Genevieve (Post-Doc, Inserm)
LAMBOUR Jennifer
(Cheffe de Projet, A4Cure)
to be recruited (IE, A4Cure)
Members of ECELLFRANCE
Immunomonitoring platform
AFONSO Georgia
(IE, UM),
DUFOURCQ-LOPEZ Emilie
(TCN, CHU)
El IDRISSI Soukayna
(Gestionnaire)
OZTURK Ekin (Stagiaire)
Immunotherapy
Dendritic cells
Treg lymphocytes
Tolerance
Arthritis
Rheumatoid arthritis
Immunomonitoring
Experimental Arthritis mouse models with clinical and immune monitoring
Treg plasticity and Immune Tolerance Breakdown in RA
Immunomonitoring of patients treated by biotherapies
Dendritic cell and Treg differentiation
The research aims of my group is focusing on:
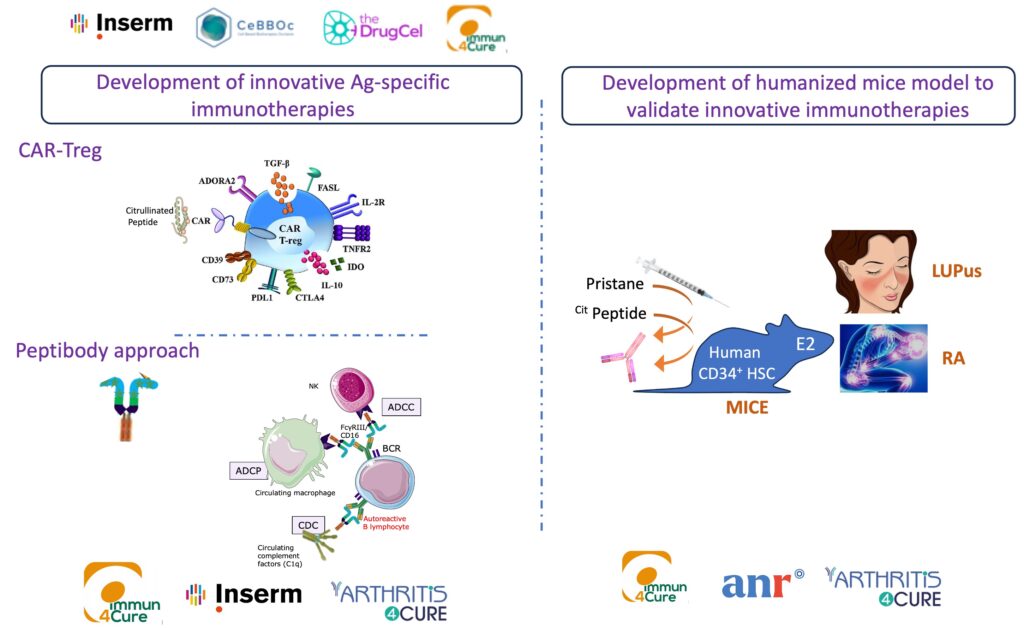
Restore immune tolerance without inducing global immune suppression using innovative antigen (Ag)-specific therapeutic molecule is the main issue of the CURE RA consortium (2018-2025), a collaborative project between 4 academic teams and the Arthritis4Cure company. As a proof of concept, we demonstrated during the last 7 years, that an innovative hybrid molecule composed with a recombinant Fc region of an Ab, “clicked” with a citrullinated peptide, is able to specifically eliminate anti-citrullinated protein antibodies (ACPA)-positive B cells in vitro and in vivo, using antibody-dependent cell-mediated cytotoxicity (ADCC) and antibody-dependent cellular phagocytosis (ADCP) mechanisms. These results allowed us to deposit 3 patents and create the A4Cure company. This innovative approach makes it possible to eliminate specific autoreactive ACPA+ B cells in RA patient. More importantly, our work opens up a new field of targeted treatment for systemic autoimmune diseases. However, the major limitation of our pilot study is that it showed in vitro and in vivo specific elimination of ACPA+ B cells and lacked substantial evidence to prove the therapeutic effects of the approach in vivo. The objective of our current projects is to advance our understanding of the role of ACPA+ B cells in the pathophysiology of RA and to validate our therapeutic innovative approach to Cure RA. The 3 aims of the project are : 1-To address the safety of the therapeutic potential in immunocompetent mice. 2- To develop ACPA dependent experimental model of arthritis and evaluate the therapeutic potential of the elimination of ACPA+ B cell. 3- To determine the efficiency and specificity of the therapeutic approach in humanized mice model and finally to develop new experimental model based on humanized mice to validate innovative molecules developed in the IHU IMMUN4Cure.
Main achievements: